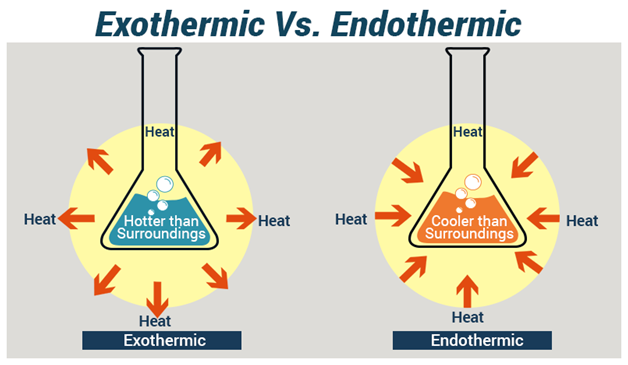
Exothermic or Endothermic Reactions, which one do you want to use for your project?
Background Information on the Available Chemicals
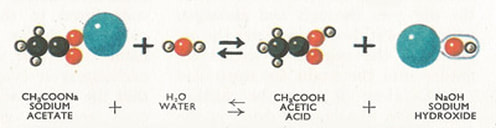
Sodium Acetate
|
Sodium acetate has a chemical formula of NaCH3COO. It doesn't have a common name. It can be made from the products of the volcano reaction. Acetic acid can be neutralized with baking soda and the water can be boiled off the resulting liquid, which will leave sodium acetate crystals. It can be used to seal concrete. It can be used in food like chips to give them a 'salt and vinegar' flavor.
|
The ingredients to make sodium acetate!
|
Sodium Chloride
|
Sodium chloride is also known as table salt or halite. The chemical formula NaCl, representing a 1:1 ratio of sodium and chloride atoms. Sodium chloride is the salt most responsible for salinity of the oceans. In its edible form of table salt, it is commonly used as a condiment and food preservative. Another major application of sodium chloride is de-icing of roadways in sub-freezing weather.
|
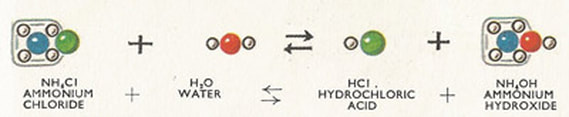
Ammonium Chloride
|
Ammonium chloride has a formula of NH4Cl and a white crystalline salt and dissolves easily in water. Sal ammoniac is a name of the natural, mineralogical form of ammonium chloride.
It is mainly used as a fertilizer for plants and a flavoring agent in some types of liquorice. In medicine, it can be used to alleviate coughing. The mineral is commonly formed when burning coal. It is also found around some types of volcanic vents. |
Ammonium Nitrate
|
Ammonium nitrate has the chemical formula NH4NO3. Its common name is Ammonium Nitrate. It is a white crystal solid and easily dissolves in water. It is mostly used in agriculture as a high-nitrogen fertilizer.
Its other major use is as a component of explosive mixtures used in mining, quarrying, and civil construction (the chemical is not explosive on its own). In nature, Ammonium nitrate is found as a natural mineral called niter in the driest regions of the Atacama Desert in Chile |
Calcium Chloride
|
Calcium Chloride is a salt with the chemical formula CaCl2. Unfortunately it doesn't have common salt name. It is a colorless crystalline solid at room temperature, and easily dissolves in water. It lowers the freezing point of water, calcium chloride is used to prevent ice formation and is used to de-ice.
The average intake of calcium chloride as food additives has been estimated to be 160–345 mg/day. Calcium chloride is permitted in food as it is considered as generally recognized as safe (GRAS) by the U.S. Food and Drug Administration. |
Calcium chloride is used in canned vegetables, in firming soybean curds into tofu. It is commonly used as an electrolyte in sports drinks and other beverages, including bottled water. The extremely salty taste of calcium chloride is used to flavor pickles without increasing the food's sodium content. Calcium chloride's freezing-point depression properties are used to slow the freezing of the caramel in caramel-filled chocolate bars. Also, it is frequently added to sliced apples to maintain texture. It can also be used in cheesemaking!